Winter is coming… It’s a scary thought, at least in Montreal. Amusingly enough, 200 years ago, this same cold was a great luxury that only the elite of societies possessed. After all, what’s cooler than a fresh cold beer? Back then, Frederic Tudor, also known as Boston’s Ice King, took advantage of this, as he shipped ice to the Caribbean in the early 1800s. Thanks to his ice business, he became one of the first American millionaires.
Unfortunately for him, in the second half of the 19th century, a better understanding of heat by scientists like Rumford and Joule led to the design of a machine that would revolutionize the world: the refrigerator. It destroyed Tudor’s ice business! In this article, we’ll present the physics of cold. Then, we’ll explain how cold machines work. Finally, we’ll talk about the Frontier of cold, absolute zero, and its extremely surprising properties, like the Bose-Einstein condensate experimentally discovered in 1995 and rewarded by a Nobel prize in 2001.
The Physics of Cold
Frenchman Antoine Lavoisier is often regarded as the father of modern chemistry. In particular, he’s known for the law of conservation of mass, according to which mass cannot be created nor loss. Rien ne se crée, rien ne se perd, tout se transforme. His study of basic elements of matter led him to postulate that heat corresponded to a fluid called caloric which pushed away particles of elements. The more caloric, the higher the volume of an element. According to this theory, called the caloric theory, cold was the absence of the caloric.
According to Lavoisier, yes. But this was about to be disproved by the American-born British physicist sir Benjamin Thompson, Count Rumford. Indeed, Rumford showed that heat could be created apparently indefinitely by friction. It could therefore not be a substance. This is also how Rumford came to visualize heat as the motion of elements. It took him 50 years to convince people though, as Lavoisier’s prestige made people trust him over Rumford. But after Lavoisier’s death during the French revolution, Rumford took this prestige away (as well as Lavoisier’s wife!).
Ironically though, the common models of heat transfer rather correspond to the caloric theory, as you can see on this figure from Wikipedia, where heat flows from the red hot disc to the blue colder one:

No! They’re great models! But they conceal the actual physics of heat. It’s like considering that species evolve without an insight into DNAs.
As hinted at by Lavoisier, fluids and solids are actually made of billions of billions of billions of tiny particles called molecules. These particles are all moving at high speed. Rumford claimed that heat was a measure of the speeds of particles. As a result, cold corresponded to fluids and solids with slow particles.
Exactly! Just like you’d have trouble staying still if you’re right at the exit of a football stadium at the end of the game, a particle can gain motion as other particles collide it. This is how heat is transfered at the microscopic level. At the macroscopic level, this corresponds to a global transfer of the average speed of particles between two objects. This is what English physicist James Prescott Joule called a transfer of energy.
Exactly! At Joule’s time, it was known that flow of heat could induce a mechanical motion. This corresponds to steam machines. However, the amazing discovery made by Joule is that things could occur the other way around, that is, mechanical motion could be converted into heat. As he made paddles rotate in water, he noticed a small increase in the temperature of water. What Joule postulated is the equivalence of the energy of motions and the energy of heat.
His idea has definitely revolutionized physics, which now postulates that the energy of any closed system is conserved. This is known as the first law of thermodynamics.
In modern science terms, heat is the average speed of particles. The speed of each particle corresponds to a kinetic energy, and these energies add up to form the thermal energy of an object. The hotter the object, the higher the thermal energy.
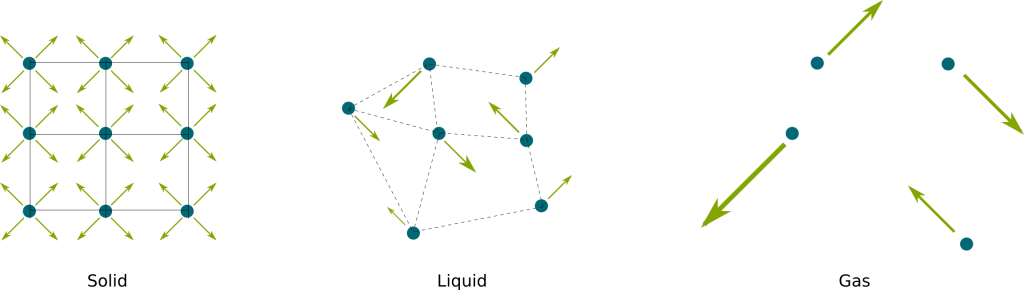
They do! In fact, they vibrate around their positions, as displayed in the following picture:
The difference between the different phases is the strength of the links between particles. As you may have guessed, when the vibrations of particles in solid increase, they may suddenly break the electromagnetic links that strongly tied them together to become liquid. Similarly, as vibrations of liquid particles increase, there may no longer be any electromagnetic link between them anymore. The breaking of these links require an additional amount of energy, called the enthalpy of phase transition.
In fact, there is a limit to the vibrations of particles in solid phase. This limit depends on the strength of the links, which depend on the type of particles and on the pressure. Now, for a given type and a given pressure, this means that the temperature of a solid object cannot exceed the temperature of the maximum vibrations of particles in solid phase. As a result, when an object is melting, the thermal energy you are giving to it will be used to break the links, because particles cannot be accelerated in the solid anymore. This means that when heat ice and when ice starts melting, ice will remain at the same temperature. At atmospheric pressure, this temperature at 0° Celsius (32° Fahrenheit).
Yes! When a liquid evaporates, the temperature of the liquid is constant. For instance, when water is boiling in your casserole at atmospheric pressure, it will remain at 100° Celsius (212° Fahrenheit). This fundamental remark is what led to the invention of refrigerators!
Refrigerators
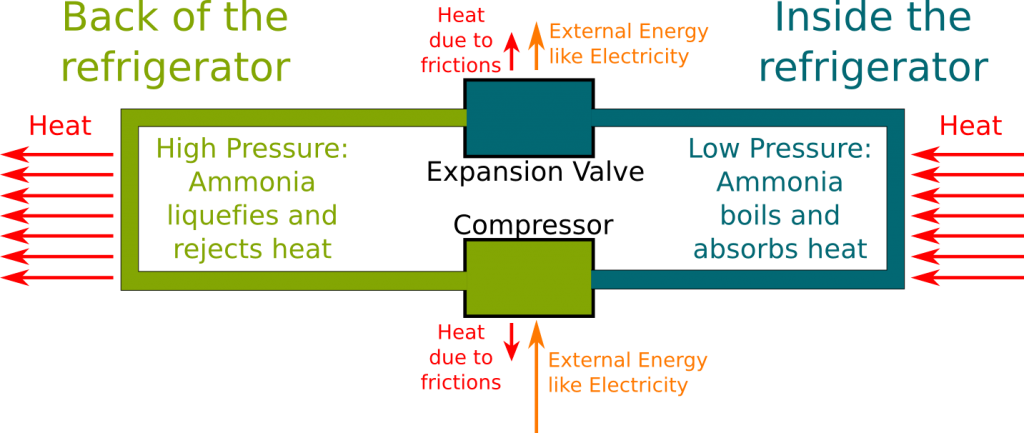
As I said, refrigerators are based on the idea that evaporating liquids remain at constant temperature.
What we have also seen is that the the boiling temperature depends on the strength of the links between particles, which in turn depends on the sort of particles. Obviously, to keep things at low temperatures, water is not the right type of particles. In fact, the sort of particles we need to use is the sort of particles which aren’t liquid anymore above the temperature we want to reach.
At atmospheric pressure, the boiling temperature of oxygen and nitrogen are respectively about −183°C (-297°F) and -196°C (-321°F). That’s a bit too cold for our refrigerators! Instead, refrigerators use ammonia (NH
Basically, yes!
To answer this question, you need to notice that the given boiling temperatures are given at atmospheric pressure. But if we increase pressure, boiling temperatures are increased too. This means that with enough pressure, the boiling temperature can reach atmospheric temperature. All we need to do to obtain liquid ammonia at atmospheric temperature is to increase pressure!
By contracting the gas of ammonia by means of pumps! This pumps can be powered by steam engines or, in the case of today’s refrigerators, by electricity!
Exactly! This is displayed in the following figure:
This is a great remark! As the ammonia condensates, it releases energy to make the links between particles. This creates heat, which is escaped by fans. The energy of cooling the refrigerator actually goes in heating the back of your refrigerator!
But that’s not all! I have put arrows of loss of heat due to frictions around the expansion valve and the compressor only, but frictions are actually occurring all along the flow of ammonia. The energy consumed by your refrigerators corresponds to these frictions.
You must be talking about the absorption refrigerators. Their principle is to heat the gas of ammonia to increase pressure and then cooling it to liquefy it as pressure is maintained high. The idea is basically the same, except that, instead of using electricity to mechanically compress ammonia, we heat it to create great pressure.
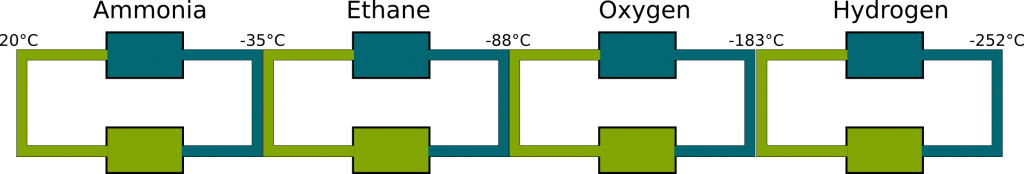
Indeed. Except that the pressures to make them liquid at atmospheric temperatures are enormous. Instead, one way of liquefying these gas is to use first ammonia to condense them at lower pressure. In fact, we could do a chain of such gases to create colder and colder objects, as displayed in the figure below:
For a long time, scientists thought that hydrogen had the lowest boiling temperature, and it was considered as the Everest of cold. In 1898, Scottish physicist James Dewar became the first to produce liquid hydrogen, after dangerous experiments based on the process described in the figure above. Indeed, the liquefaction of hydrogen involved extremely low temperatures and high pressure which led to frequent explosion of tubes. When he finally did it, while he was expecting prestigious awards, an element with even lower boiling temperature was discovered: helium.
There was one big problem… He didn’t have any helium! And because of the bad way he treated his collaborators, some of them refused to give him helium! Eventually, he was preceded by Dutch physicist Heike Kamerlingh Onnes, who, as opposed to Dewar, got awarded the Nobel prize for his exploit!
Liquid helium is pretty cool, as it boils at -269°C.
Towards Absolute Zero
As we go into the world of cold, amazing properties occur. In particular, an extremely weird phenomenon was unveiled: superconductivity.
It’s the property of having no resistance to electric current. As a result, extremely powerful currents can go through superconductive matters, and they would not heat nor lose any energy. This can be used to create major magnetic vibrations. The consequences of creating strong magnetic fields would be incredible! According to Michio Kaku in his book about the physics of the future, magnetism will be ruling the 21st century, just like electricity has ruled the 20th century.
Today, we already use magnetism to scan brains with MRI. Kaku envisioned a future where computers could directly read through our brains and answer our desires. It will be like having telekinetic powers. Another great application regards transports, as displayed in the following video:
But that’s only the first step into the wonders of cold. Scientists have been racing to achieve the coldest states of matters.
Well, temperature corresponds to the motion of particles…
That’s the idea! This limit is called absolute zero. It’s about -273.15°C. This has led scientists to define a new scale of temperature, called Kelvin, which is equal to 0 at absolute zero, and increases at the same rate as degrees Celsius. For instance, boiling temperature of helium at atmospheric pressure is about 4 Kelvins.
Now, intuitively, you may think that absolute zero corresponds to absence of motion. But in fact, at this temperature, there are a few quantum fluctuations, as explained by physicists of the University of Nottingham on Sixty Symbols:
No. But that’s because it’s theoretically impossible to get to this temperature! In theory, we could get as close to absolute zero as we want, but we could never get to it… at least not in a finite time! But they have come extremely close, at less than a millionth of degree (a few nanoKelvins)!
You could obtain lower boiling temperature by decreasing pressure. If you do this, then, at 2 Kelvin, you can reach a new state of matter called superfluid. This state of matter is extremely weird, as it has zero viscosity and infinite thermal conductivity. It only exists at extremely low temperature, like for liquid helium, or extremely high pressure, like in neutron stars:
But to make even colder cold, you need new technics than liquefying gases.
Mainly, laser cooling!
Not necessarily… The idea of laser cooling is based on two major principles. First, particles are sensible to specific light frequencies only. They don’t interact with other frequencies. To find out why, you’ll have to get through my article on the dynamics of the wave function in quantum mechanics. Second, an observer which goes in the opposite direction of light will see the light having a higher frequency than a standby observer. This is the Doppler effect, which explains the sound of ambulances.
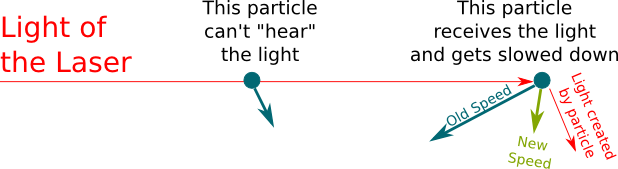
Laser cooling consists in sending lights with frequencies such that, because of the Doppler effect, only fast moving particles in the opposite direction of the laser receive them. Particles then receive the momentum of lights in the opposite direction of their motion, which slows them. They then lose the momentum of the lights they eject which is likely not to be in the opposite direction of their motion, which means that, in average, they don’t speed up. That’s why, overall, they slow down. This is displayed by the following figure:
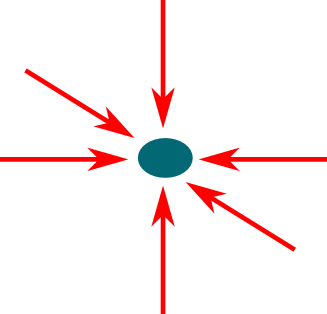
Indeed. That’s why we need several lasers coming from all directions, as displayed below:
It did. But even better results have been obtain by adding magnetic fields.
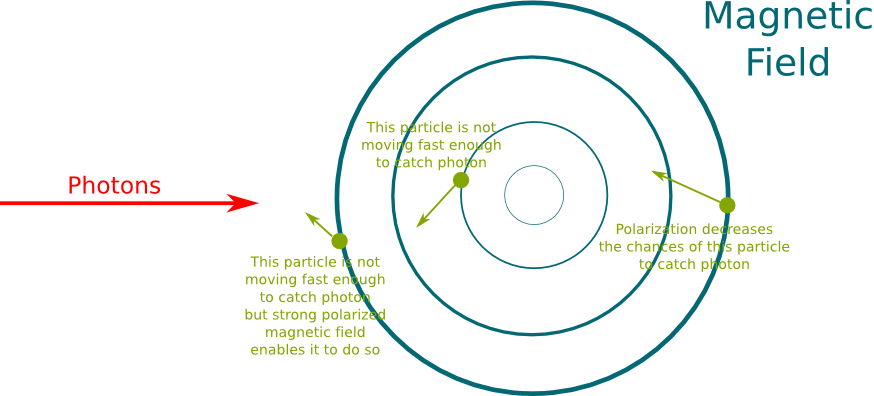
This is due to the Zeeman effect. In presence of a magnetic field, depending on polarization, the frequency of light that a particle can absorb gets modified. As a result, by tuning the right polarization of magnetic field, and increasing its strength far from the central area, we can increase the chance of particles moving away to absorb a laser photon which would kick them back towards the central area. This is displayed in the following figure.
Now, slow moving particles will be more easily kicked back towards the centre, as they only need to capture a few photons for that. However, fast moving particles will be moving away despite capturing photons. This means that the magnetic field is also a way to get rid of particles moving too fast. And this is even more the case as its strength gets lowered. By doing so, lower and lower temperatures can be obtained. Until suddenly…
The answer to this question was given by Satyendra Nath Bose and Albert Einstein in 1924-25! But it was a theoretical answer, and Einstein never thought it could be observed. So when the ideas of laser and magnetic cooling appeared, scientists have raced to observe Bose and Einstein’s prediction.
The Bose-Einstein condensate! It’s an extremely weird state of matter, where particles group together to form a unique large quantum object.
Yes, they have! In 1995, after decades of research, Eric Cornell and Carl Wieman created for the first time, maybe in all of our universe, a Bose-Einstein condensate. They preceded Wolfgang Ketterle by a few months, but all three have been rewarded with the Nobel Prize in 2001.
Applications for real life are not to be expected tomorrow, but the Bose-Einstein condensate might one day prove itself extremely useful.
It could be used to design quantum computers. These computers, instead of working with bits, manipulate wave functions which contain an awfully lot more of information. This would enable complex operations to occur simultaneously. But it also requires computer scientists to totally rethink the way algorithms work. For one thing, they are necessarily probabilistic algorithms. I talk about quantum algorithms in this article. Another application is quantum cryptography.
Yes… But this doesn’t mean that low thermal energy has been reached. Rather, this is due to the weird actual definition of temperature as the variation of entropy per energy added. That’s what’s explained in the following short video by Minute Physics:
Let’s Conclude
For long, people feared cold. It was the absence of the warmth inside houses. But its mastery proved very helpful. Think about skyscrapers for instance. At high heights, winds are so strong that windows can’t open. There would be no way of ventilating skyscrapers. And no way of regulating their temperatures. But thanks to air conditioning it’s now possible, and people can work in a nice environment, even on tops of buildings of Manhattan, Hong Kong or Singapore.
The journey towards cold has been extremely exciting, quite similar to a hike to the summits of mountains. I love this documentary and I strongly recommend you to check it out. It shows science as a dramatic adventure through the wonders of Nature. More importantly, it shows how awesome science is!




Very nice blog post. I certainly love this website. Stick with it!