In the 20th century, science has unveiled an incredible world that even the most daring science-fiction writers could have never even thought of. This world was so surprising that it completely puzzled the most brilliant minds of the century, from Albert Einstein to Richard Feynman, from Niels Bohr to Paul Dirac, from David Hilbert to Werner Heisenberg or from Erwin Schrödinger to Wolfgang Pauli. Apparent paradoxes kept being found, but they all ended up to be caused by mistaken common sense. Meanwhile, the central question of the measurement problem around which quantum mechanics is constructed still has no commonly accepted explanation…
The basic ideas of quantum mechanics are definitely troubling. But they are not that complicated. The major difficulty is to get rid of common sense because common sense makes a lot of mistaken assumptions. This is why learning quantum mechanics is an extremely humiliating and fruitful journey to undertake. Also, quantum mechanics has terrible philosophical implications. That’s why I invite you to embark on the adventure of the unveiling of quantum mechanics. To get you excited, let’s start with an extract from the Fabric of the Cosmos: Quantum Leap, by NOVA and hosted by Brian Greene:
Wave-Particle Duality
For long, physicists have been debating about the true nature of light. While Newton imagined it made of small particles, this rather intuitive concept was suddenly shattered by the double slit experiment realized by Thomas Young in the early 1800s. In this article we will almost only focus on this experiment and its numerous variants, as, according to Nobel prize winner Richard Feynmann, [it] has in it the heart of quantum mechanics. In reality, it contains the only mystery.
A beam of light was sent on two tiny close holes, before being captured on a screen behind. The screen then displayed a troubling interference pattern, that is, a succession of dark and bright areas. The following video by Veritasium displays the experiment with so simple tools that you can do it yourself:
This experiment is usually rather done with lasers, but Derek Müller’s box enables to take it to the street in a pretty cool way.
Indeed. To explain this phenomenon, the wave theory of light was introduced, with James Clerk Maxwell’s theory of electromagnetism as the golden age of the theory. In this setting, light was seen as an electromagnetic wave.
OK. First, let me talk about fields. A field is something that fills up space. More precisely, at any point in space, the field takes some value. The concept of field is illustrated in the following extract of a video by Minute Physics:
Now, if, at some point of the field, values are high, then there is a perturbation of the field at this point. This perturbation can then propagate, like waves on water. A wave is a propagating perturbation of a field.
Yes! This enabled to explain a lot of other phenomenons. But then came the discovery of the photoelectric effect.
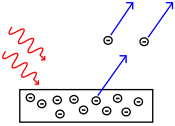
In some specific setting, lighting a metal could induce an electrical current. What was troubling is that the electrical current was induced if and only if the wavelengths of light were small enough. If you used red light, there was no current. But if you used blue light, there was. This appeared like a discontinuous phenomenon which was incompatible with the continuity of Maxwell’s equations.
In one of his 4 earthshaking papers of 1905, Albert Einstein explained the discontinuity by the fact that light could actually be thought of as a composition of elementary particles called quanta of light or photons! He justified this surprising assumption by the fact that the energy distribution of light was very similar to the energy distribution of particles of a gas. Another evidence supporting his idea was the discovery of spectral lines of emission and absorption.
I know! Yet, this didn’t prevent Einstein from receiving a Nobel prize for his idea of photons.
We’ll get to this! But for now, let’s keep confusing ourselves with another weird experiment. As opposed to light, it was thought that all matter was made of atoms, and that these atoms were made of protons, neutrons and electrons. In particular, electrons were considered as elementary indivisible particles. Indeed, scientists managed to isolate them one at the time…
But they then tried Young’s double slit experiment with beams of electrons instead of beams of light, and found interference patterns! This is displayed in the following figure from the Northern Arizona University website:

This means that, just like photons, electrons appear to have both particle and wave properties! This is what scientists called the wave-particle duality, and is often interpreted as particles sometimes behaving like waves. In fact, quantum field theory even suggests that forces also are both waves and particles, as explained in Thibault’s article.
I totally agree! So let’s better understand what’s going on!
Wave Function
The solution provided by many scientists in the 1920s and 1930s was to think of matter and light as a collection of elementary entities. But these elementary entities, which we call photons, electrons or anything else, are not actually particles. Rather, each entity is a wave. However, physicists have now got used to calling these elementary entities particles, but you need to keep in mind that these are not to be thought of as point objects but rather as waves. Let me rephrase the scientists’ idea of that time because it’s very important: All things are made of elementary waves.
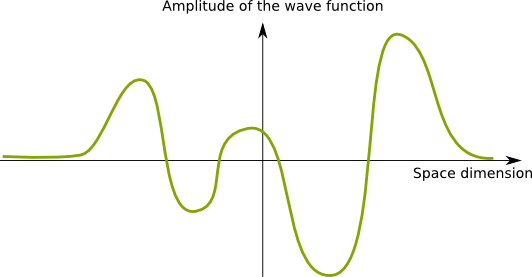
I know! The following figure displays a 1-dimension wave function, that maps locations with the amplitude of the wave. Keep in mind that the following wave function represents one particle only.
Yes, because elementary entities are waves! These waves can add up to be constructive or destructive, creating interference patterns.
Yes too, because beams of electrons are actually a collection of elementary waves known as electrons.
This enabled to account for the photoelectric effect as well. The current is induced if electrons can capture an elementary wave which is energetic enough to make them leave the orbit of the atoms. The captured elementary wave is a photon with enough energy.
It surely does! But there are more troubling experiments I haven’t mentioned yet… Because electrons can be isolated, it’s possible to do the double slit experiment by shooting them one at a time. We can then visualize where they land on the screen. Let’s recapitulate all the double slit experiments we have discussed so far with the following video of Dr Quantum:
Each electron lands at one particular location on the screen. But the locations where they arrive are not all the same! This is very weird, since all electrons are similar. More precisely, they are described by identical wave functions. And yet, they all end up at different location!
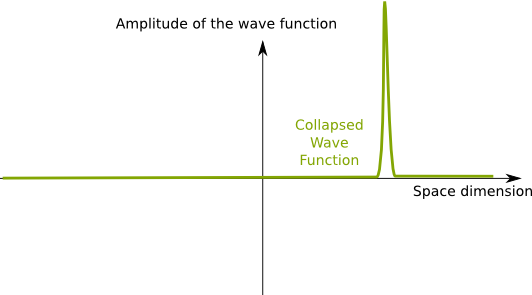
Physicist Max Born proposed that, when measured, the waves collapse in a inherently random way. This means that they become very concentrated at a relatively precise location. This is usually interpreted as them becoming particles. But to really understand it, you should keep in mind that a collapsed wave is a very localized wave, rather than a point particle. A localized wave is almost nil almost everywhere except around the location of the wave:
Once collapsed the wave then resumes its propagation. But more than the way waves look, what needs to be stressed is the fact that the collapse of the wave functions occurs in a inherently random way. This means that two identical experiments yield different results!
These were questions that even Albert Einstein couldn’t handle, as he famously refuted quantum theory by stating that God doesn’t play dice with the world. Yet, experiments after experiments, over almost 100 years, this quantum effect has been proven again and again.
What I haven’t told you yet is that, behind this seemingly quantum unpredictability, there actually is amazing well-defined world of probabilities. Even though the results of experiments could not be predicted, the probabilities of the outcomes could be computed with incredible precisions.
Let’s reconsider the double slit experiment with one electron at a time. As more and more electrons were sent, an interference pattern appeared, as if all were sent simultaneously. What physicists realized is that this interference pattern was revealing the probabilistic distribution of the outcomes of the single electron experiment. Yet, this interference pattern also corresponded to the wave functions!
Precisely! From the elementary waves that form all things are deduced the probabilistic distributions of where they collapse! More precisely, the square of the norm of the value of the wave function at a point is the probability of finding the collapsed wave there once we measure it. This is explained in the following extract from The Fabric of the Cosmos: Quantum Leap:
You need to consider the waves to understand constructive and destructive interferences. Also, waves contain information about energy and momentum of particles. But more importantly, as explained in this more advanced article, the dynamics of the wave is precisely described by Schrödinger equation. But finally and more importantly, according to quantum mechanics, the true nature of particles is being waves. What most popularizing videos call particles should rather be called localized waves. If you keep this in mind, I think you’ll understand quantum mechanics much better.
The Measurement Problem
It’s time for us to get to the most troubling part of quantum physics. Let’s reconsider once again the double slit experiment. As physicists tried to understand how the electrons were moving, they decided to add detectors to observe slits through which electrons were moving. The result was profoundly shocking, as explained in this extract from The Quantum Universe:
When slits were observed, there was no longer any interference pattern! Everything suddenly occurred as if electrons had been moving in straight lines all along. This was absolutely shocking: The outcome was modified by the mere fact that we were observing electrons along the way!
Detectors showed that electrons were moving through one of the slits, and one only! Recall that when we weren’t observing slits, the wave was spread and went through both slits.
Yes, exactly! In fact, if we consider that particles are always waves that simply collapse randomly for any measurement, then we can understand what is happening. As we measure the particle at the slits, it collapses and becomes very localized. Because the size of the slit is larger than the spreading of the collapsed wave function, it’s good approximation to say that the particle only goes through one slit. Now, because the wave function collapsed, it’s no longer the same. Thus, it will not behave as if it hadn’t collapsed. This means that the result will no longer be the same as if we hadn’t observed particles at the slits. That’s why observation of slits affects the outcome of the experiment.
I know! But wait! There’s more. New technologies have enabled to delay the observation of slits, as explained below:
And yet, even when the observation of slits was delayed, things occur as if the observation of slits wasn’t delayed: There was no interference pattern!
I know. This is extremely troubling! This leads us to the most fundamental misunderstood question of quantum physics, known as the measurement problem. Shortly put, it consists in asking: What’s a measurement? Out of context, it would sound like a technical detail. Yet, as we have been discussing it, measurements are parts of the laws of physics, as they affect the outcomes of experiments. And yet, we don’t even know what they really are.
In 1935, Nobel prize winner Erwin Schrödinger introduced one of science’s most famous thought experiment to illustrate how much we don’t understand measurement (although I’ve heard that his goal was rather to ridiculize quantum physics).
Yes! It consists in putting a cat in a box with a radioactive atom and poison. If the atom decays then the poison is released and the cat dies. The box is left for a minute. This gives the atom a fifty-fifty chance to decay. Now, if don’t equip the box with any measurement device, then it is in a quantum state. This implies that the cat is both dead and alive, as displayed in the following video from The Open University.
No! Reconsider the double slit experiment. When we observe slits, the electron is indeed in the left or right slit. But if we don’t observe slits, it is both in the left and right slits, because the wave function spreads over the two slits. The two cases are very different, as we can see in the results of the experiments. Similarly, here, the cat isn’t dead or alive. It is both dead and alive. And until measurement, that is, opening the box and checking, it is in this superposition of states.
Well, that’s a troubling question you’re asking here… This is precisely the measurement problem! And, shortly put, there is no accepted explanation for it.
We’ll get to this eventually. But first, let me show you the measurement problem at its worst: entanglement.
Let’s reconsider Schrödinger’s cat. Assume we could now separate the radioactive atom and the cat, after they have been put together for a minute. Let’s keep the radioactive atom with us on Earth, while we send the cat far far away, in some distant galaxy. Now, if we still haven’t made any measurement of the cat nor the atom, both are in a quantum superposition state. The cat is both dead and alive, while the atom both has and hasn’t decayed. But the fates of the cat and of the atom are linked. When we measure the state of the atom, quantum mechanics says that it will instantaneously affect the state of the cat.
Yes! And if the atom hasn’t decayed, then the cat instantly lives! It’s as if the information about our measurement instantaneously affected the state of the cat.
He did! In his theory of relativity, Einstein proved that nothing travels faster than light! Worse than that, he proved that simultaneity didn’t even exist, as it depended on the observer. Therefore, entanglement seems to totally contradict his theory! This was unacceptable for Albert Einstein, who famously referred to this phenomenon as spooky action at a distance.
What you’re saying corresponds to saying that the cat is dead or alive rather than dead and alive. That’s what Einstein suggested. After decades of endless debates, Irish physicist John Bell proposed an experiment to settle the arguments. This experiment was then improved by Alain Aspect who separated the cat and the atom far enough so that there had to be a faster-than-light communication. This is explained in the Fabric of the Cosmos: Quantum Leap.
The experiments showed that Einstein was wrong. The cat and the atom were actually in a quantum superposition state, and there was an actual spooky action at a distance.
Weirdly enough, it appears so. Applications are amazing! Perfectly secure cryptography can be achieved, as explained in Scott’s article on quantum cryptography. Even crazier, as shown later in the Fabric of the Cosmos: Quantum Leap, teleportation devices are currently being constructed based on entanglement. So far they’ve teleported photons, but at some point in the future, they might teleport people! The idea is based on the possibility of influencing the quantum state of the atom to increase the chances of eventually measuring it not-decayed. This instantaneously affect the state of the cat and increases its chances of being alive, when we will have measured the state of the atom.
In a talk at Google, Ron Garret provides a better understanding of entanglement. As he says, what entanglement really is is nothing less than a measurement. Entanglement isn’t hard to understand, provided that we really understand what measurement is. All the difficulty of the understanding of quantum physics therefore boils down to the big question I mentioned earlier…
Yes! That’s the big question of quantum mechanics! Let me recall what we have found out about measurement in this article. Measurement implies a collapsing. But it’s not only about the collapsing of a wave function. Rather, it’s a collapsing of a collection of wave functions throughout time (as shown by the double slit experiment with delayed observation of slits) and space (as proved by entanglement). This means that, because of this property of measurement in quantum mechanics, our universe is inherently a non-local, and everything seems connected.
It’s very hard to grasp the concept of measurement. Any explanation of it must involve a ground-breaking twist of our vision of the world. In fact, one of the rare ideas which might make sense is a terribly shocking one. Einstein once said: I like to think that the moon is there even if I am not looking at it. But Einstein’s view has been completely shattered by the measurement problem of quantum mechanics. Instead, many physicists claim that there is no out there out there, and that reality only exist when we look at it. I’ll leave you with the explanation of Dr Amit Goswami, in this extract from The Holographic Universe:
To face the measurement problem, a new concept of reality is probably needed, and Hawking’s idea of model-dependent realism may well help us. But it’s far from being an accepted concept to this day. Now, things may not be as weird as what I’ve just presented here. As explained by the theory of decoherence, the apparent collapse of the wave function may be just a side effect of the Schrödinger equation applied to a great number of particles. This is what I’ve explained in this extract of my talk More Hiking in Modern Math World:
Let’s Conclude
By assuming that all things are made of elementary waves, and by using equations of quantum mechanics to study the dynamics of these waves, physicists have been incredibly successful in explaining a very large range of weird experimental observations. This simply makes quantum mechanics the most tested and yet not refuted theory science has ever produced. And its applications to technologies are overwhelming! In particular, the invention of the transistor, which has then led to the explosion of new electronic and telecommunication technologies, has been the result of the understanding of quantum mechanics.
What a crazy world our world seems to be! You’re probably deeply confused. I know I am. Let me reassure you by a few more quotes by Nobel prize winners:
- Anyone not shocked by quantum mechanics has not yet understood it. (Niels Bohr)
- If that turns out to be true, I’ll quit physics. (Max von Laue, on the wave properties of electrons)
- Had I known that we were not going to get rid of this damned quantum jumping, I never would have involved myself in this business! (Erwin Schrödinger)
- Not only is the Universe stranger than we think, it is stranger than we can think. (Werner Heisenberg)
- I think I can safely say that nobody understands quantum mechanics. (Richard Feynman)
- God not only plays dice, He also sometimes throws the dice where they cannot be seen. (Stephen Hawking, not a Nobel prize winner, on black holes)
Sure. The essence is that everything is made of elementary waves. Their dynamics either corresponds to Schrödinger’s deterministic equation, or to a probabilistic collapsing. The latter one refers to the measurement problem. Although the probabilistic nature of its outcome is very well described, the cause of its occurrence is still highly misunderstood. This is partly because of its non-locality through space and time, and partly due to the lack of definition of the concept of measurement. If you want to go further in the understanding of quantum mechanics, read my article on the dynamics of wave functions!
Hum… No. Quantum mechanics seems incapable to include gravity. Although Paul Dirac managed to include Einstein’s theory of special relativity, making things work with Einstein’s general relativity is still mainly considered to be an open problem. Physicists are still in a quest to a unified theory of everything. The best candidate so far is string theory, but it’s still questioned by plenty of scientists.

Leave a Reply